Smarter Learning. Better Performance.
Optimize employee performance with data‑driven workplace learning technologies.

Learning Pool is changing how global businesses solve today’s biggest employee performance challenges
We’re doing it with data-driven learning technologies that apply insights into who a learner is, what they know, and what they need to do. By aligning learning to individual needs and business objectives, we make workplace learning experiences smarter.
Trusted by 1,500+ global companies
What our customers say
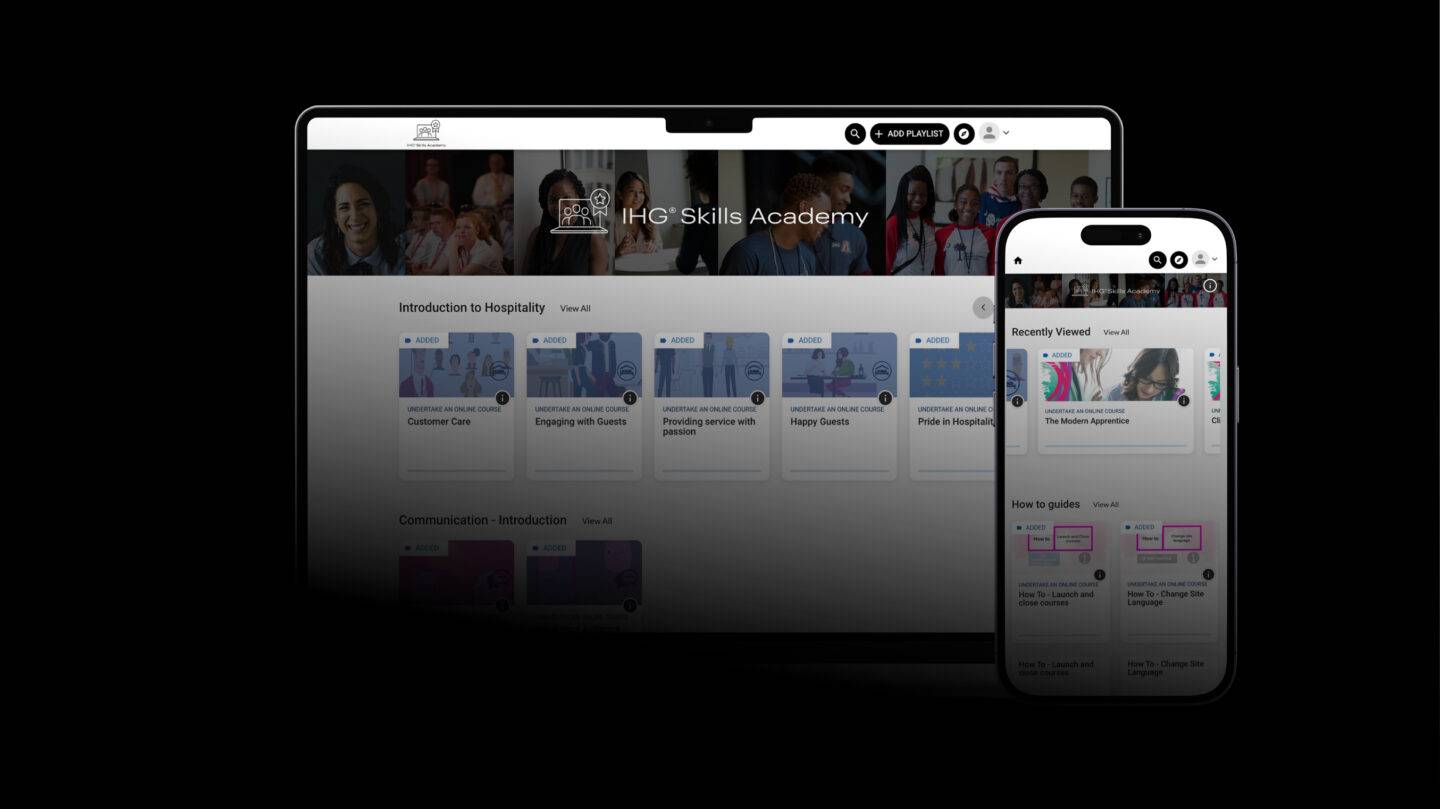
A truly exceptional story
Intercontinental Hotels Group
Industry leader
0
million active learners
0 +
customers worldwide
0 %
recommendation rate
Industry awards






Got an employee performance challenge to solve?
Get in touch to discover how our portfolio of learning solutions can help